|
|
1960
|
Detection of simian vacuolating agent 40 (SV40) as a contaminant of poliovaccines
|
Sweet BH, Hilleman MR (1960) Proc Soc Exp Biol Med 105:420-427
|
|
|
1962
|
SV40 plaque assay
|
Stinebaugh S, Melnick J (1962) Virology 16:348-349
|
|
|
1962
|
Discovery that SV40 is oncogenic in hamsters
|
Eddy BE et al (1962) Virology 17:65-75; Girardi AJ et al (1962) Proc Soc Exp Biol Med 109:649-660
|
|
|
1964
|
Detection of SV40 T-antigen expression in SV40 tumor cells and infected cells
|
Black PH et al (1963) Proc Natl Acad Sci USA 50:1148-1156; Rapp F, Butel JS, Melnick JL (1964) Proc Soc Exp Biol Med 116:1131-1135; Rapp F et al (1964) Proc Natl Acad Sci USA 52:1138; Rapp F et al (1964) Proc Natl Acad Sci USA 52:1138-1142
|
|
|
1968
|
SV40 DNA is integrated in transformed cells
|
Sambrook J et al (1968) Proc Natl Acad Sci USA 60:1288-1295
|
|
|
1971
|
Replicating SV40 genomes contain closed circular DNAs
|
Jaenisch R, Mayer A, Levine A (1971) Nat New Biol 233:72-75
|
|
|
1971
|
Cleavage of SV40 DNA by restriction enzyme and first physical map of a DNA molecule
|
Danna K, Nathans D (1971) Proc Natl Acad Sci USA 68:2913-2917; Morrow JF, Berg P (1972) Proc Natl Acad Sci USA 69:3365-3369
|
|
|
1972
|
First artificial ligation of foreign DNA into SV40: Lambda phage genes and the galactose operon of E. coli are cloned into SV40
|
Jackson DA, Symons RH, Berg P (1972) Proc Natl Acad Sci USA 69:2904-2909
|
|
|
1972
|
SV40 DNA replication is bidirectional
|
Danna KJ, Nathans D (1972) Proc Natl Acad Sci USA 69:3097-3100; Fareed GC, Garon CF, Salzman NP (1972) J Virol 10:484-491
|
|
|
1973
|
Description of adenovirus-SV40 hybrids
|
Lewis AM Jr (1973) Biohazards in biological research. Cold Spring Harbor Laboratory Press, New York
|
|
|
1975
|
Large T-antigen required for maintenance of cell transformation
|
Brugge JS, Butel JS (1975) J Virol 15:619-635; Tegtmeyer P (1975) J Virol 15:613-618
|
|
|
1975
|
The SV40 genome exists as a minichromosome
|
Griffith JD (1975) Science 187:1202-1203
|
|
|
1977
|
Identification of T-antigen as early gene A product
|
Rundell K et al (1977) J Virol 21:636-646
|
|
|
1977
|
SV40 can cause progressive multifocal leukoencephalopathy in rhesus monkeys
|
Holmberg CA et al (1977) J Infect Dis 136:593-596
|
|
|
1978
|
Determination of SV40 DNA sequence; first full genomic sequence of a virus that affects eukaryotes
|
Fiers W et al (1978) Nature 273:113-120; Reddy VB et al (1978) Science 200:494-502
|
|
|
1978
|
Spliced SV40 messenger RNAs
|
Berk AJ, Sharp PA (1978) Proc Natl Acad Sci USA 75:1274-1278
|
|
|
1979
|
SV40 large T-antigen binds p53
|
Lane DP, Crawford LV (1979) Nature 278:261-263; Linzer DI, Levine AJ (1979) Cell 17:43-52
|
|
|
1980
|
Propagation of cloned, infectious SV40 DNA in bacteria
|
Peden KW et al (1980) Science 209:1392-1396
|
|
|
1981
|
A 72-bp sequence in SV40 is a strong enhancer of transcription
|
Banerji J, Rusconi S, Schaffner W (1981) Cell 27:299-308; Moreau P et al (1981) Nucleic Acids Res 9:6047-6068
|
|
|
1981
|
Monoclonal antibodies against SV40 tumor antigens
|
Deppert W, Gurney EG, Harrison RO (1981) J Virol 37:478-482; Harlow E et al (1981) J Virol 39:861-869
|
|
|
1983
|
Sp1 binds to the SV40 early promoter
|
Dynan WS, Tjian R (1983) Cell 32:669-680; Dynan WS, Tjian R (1983) Cell 35:79-87
|
|
|
1983
|
Repeated GC-rich motifs are in the SV40 promoter
|
Everett RD, Baty D, Chambon P (1983) Nucleic Acids Res 11:2447-2464
|
|
|
1984
|
Replication of SV40 DNA in vitro
|
Li JJ, Kelly TJ (1984) Proc Natl Acad Sci USA 81:6973-6977
|
|
|
1984
|
Large T-antigen induces tumors in transgenic mice
|
Brinster RL et al (1984) Cell 37:367-379; Hanahan D (1985) Nature 315:115-122
|
|
|
1984
|
Discovery of nuclear localization signal on SV40 T-antigen
|
Kalderon D et al (1984) Nature 311:33-38; Lanford RE, Butel JS (1984) Cell 37:801-813
|
|
|
1988
|
SV40 large T-antigen binds pRB
|
DeCaprio JA et al (1988) Cell 54:275-283
|
|
|
1988
|
Mapping of cytotoxic T cell epitopes on T-antigen
|
Anderson RW et al (1988) J Virol 62:285-296; Tanaka Y et al (1988) Virology 162:427-436
|
|
|
1989
|
SV40 T-antigen binds heat shock protein
|
Sawai ET, Butel JS (1989) J Virol 63:3961-3973
|
|
|
1991
|
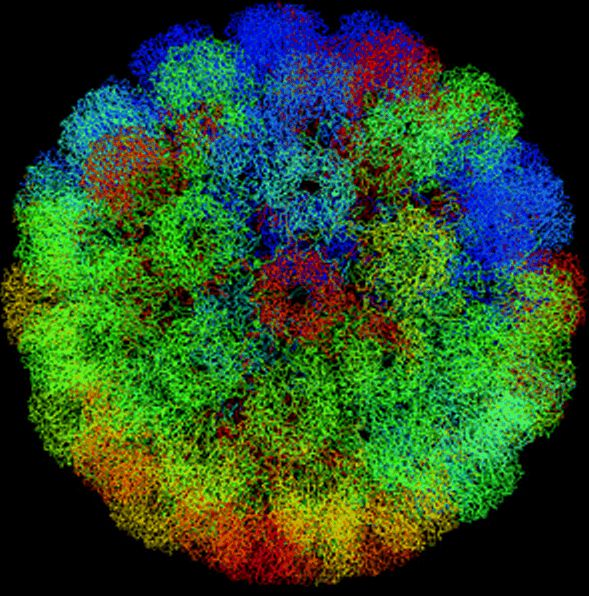
Structure of SV40 virions at 3.8- Å resolution
|
Liddington RC et al (1991) Nature 354:278-284
|
|
|
1992
|
Cell surface receptors for SV40 are MHC class I molecules and GM1 ganglioside
|
Breau WC, Atwood WJ, Norkin LC (1992) J Virol 66:2037-2045; Tsai B et al (2003) EMBO J 22:4346-4355
|
|
|
1992
|
Association of SV40 DNA with pediatric brain tumors
|
Bergsagel DJ et al (1992) N Engl J Med 326:988-993
|
|
|
1992
|
Genetic analysis of SV40 in brains and kidneys of macaque monkeys
|
Ilyinskii PO et al (1992) J Virol 66:6353-6360
|
|
|
1993
|
Discovery of 17 kDa SV40 tumor antigen
|
Zerrahn J et al (1993) EMBO J 12:4739-4746
|
|
|
1994
|
Association of SV40 DNA with human mesotheliomas
|
Carbone M et al (1994) Oncogene 9:1781-1790
|
|
|
1996
|
SV40 enters cells through caveolae
|
Anderson HA, Chen Y, Norkin LC (1996) Mol Biol Cell 7:1825-1834
|
|
|
1997
|
Major histocompatibility class I molecules mediate association of SV40 with caveolae
|
Stang E, Kartenbeck J, Parton RG (1997) Mol Biol Cell 8:47-57
|
|
|
1998
|
Genetic heterogeneity of SV40 in immunocompromised monkeys
|
Lednicky JA et al (1998) J Virol 72:3980-3990
|
|
|
1998
|
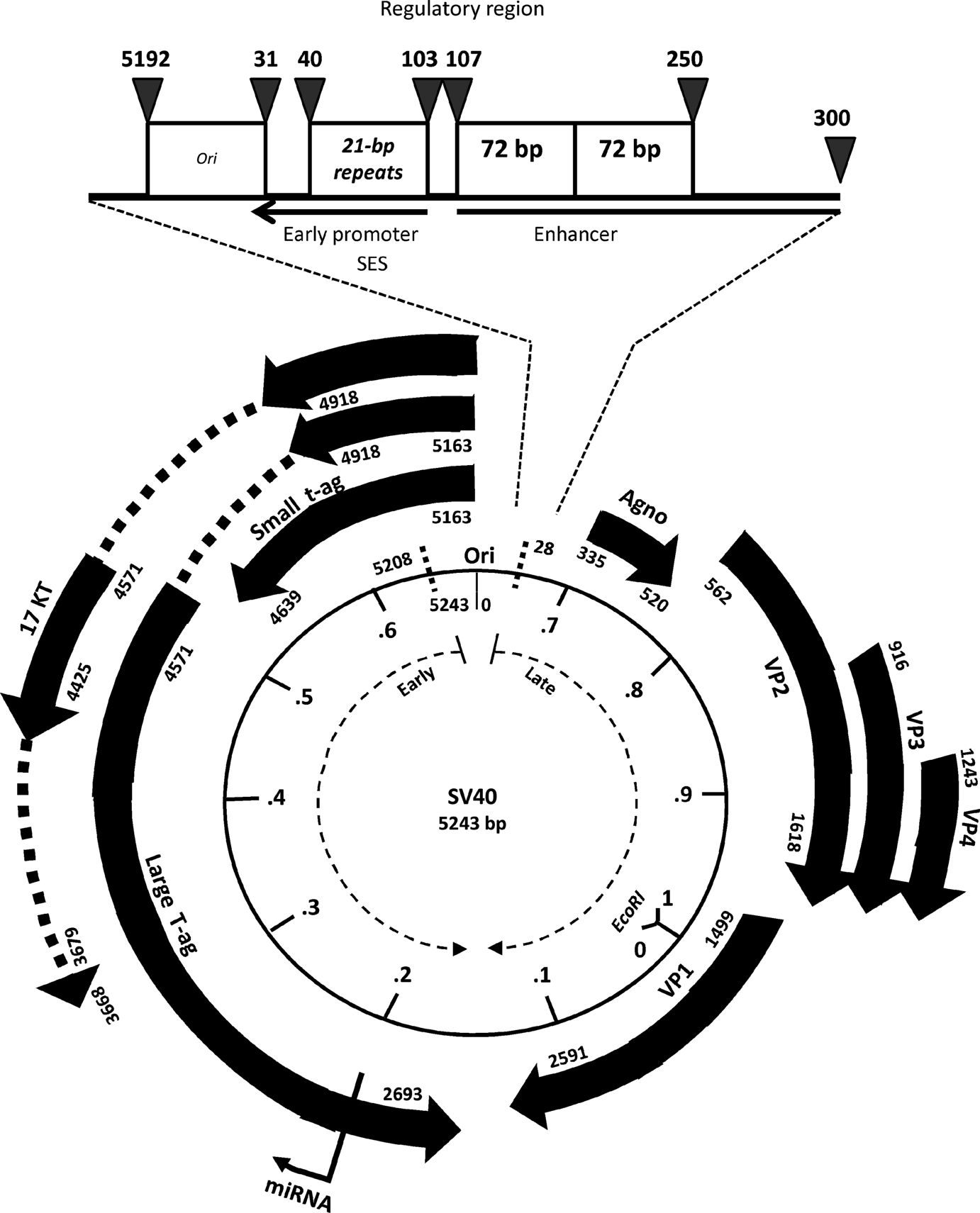
Description of the SV40 encapsidation signal (SES)
|
Gordon-Shaag A et al (1998) J Mol Biol 275:187-195
|
|
|
2001
|
SV40 regulatory region structural diversity
|
Lednicky JA, Butel JS (2001) Semin Cancer Biol 11:39-47
|
|
|
2002
|
SV40 can alter three of five cellular pathways, the disruption of which is necessary for transformation of human cells
|
Hahn WC et al (2002) Mol Cell Biol 22:2111-2123
|
|
|
2003
|
Crystal structure of T-antigen helicase domain
|
Li DW et al (2003) Nature 423:512-518
|
|
|
2004
|
Phylogenetic grouping of SV40 strains
|
Forsman ZH et al (2004) J Virol 78:9306-9316
|
|
|
2005
|
Discovery of SV40 microRNAs
|
Sullivan CS et al (2005) Nature 435:682-686
|
|
|
2005
|
Use of SV40-derived gene therapy vectors
|
Strayer DS et al (2005) Curr Gene Ther 5:151-165
|
|
|
2007
|
SV40 T/t-antigen signature from transgenic models predictive of human carcinomas with poor prognosis
|
Deeb KK et al (2007) Cancer Res 67:8065-8080
|
|
|
2008
|
Influence of viral regulatory region on SV40 tumor induction
|
Sroller V et al (2008) J Virol 82:871-879
|
|
|
2009
|
Cell-type specific global gene expression regulated by T-antigen in SV40-transformed mouse cells
|
Cantalupo PG et al (2009) Virology 386:183-191
|
|
|
2009
|
Seroepidemiology of polyomaviruses in humans
|
Kean JM et al (2009) PLoS Pathogens 5:e1000363
|
|
|